
- Home
- Test Overview
- Clinical Validation
- Order the Test
- About Us
- Contact
DecisionDx-EC
Overview
DecisionDx-EC:
Enables More Targeted Use of Therapy
Predicts extreme resistance to pre-operative chemoradiation therapy in esophageal cancer patients
The DecisionDx-EC test was discovered at The University of Texas MD Anderson Cancer Center (MD Anderson) as a way to predict likelihood of response to neoadjuvant, or pre-operative, chemoradiation therapy (CTRT) in individual patients with esophageal adenocarcinoma. The test assesses three protein biomarkers from diagnostic biopsy tissue to provide validated biological information to help design personalized treatment plans. Initial clinical validation was completed at MD Anderson and presented at the annual meeting of the American Society of Clinical Oncology in 2011.
Castle Biosciences, Inc. exclusively licensed this technology and recently completed a multi-center effort to technically and clinically validate the proprietary test. This multi-center clinical validation found that the DecisionDx-EC test was able to predict extreme resistance to CTRT with a high degree of accuracy. The test is only available through Castle Biosciences.
Today, treatment plans for esophageal cancer, based upon National Cancer Comprehensive Network (NCCN) guidelines, recommend pre-operative CTRT for tumor shrinkage and stabilization as well as improved overall survival. Unfortunately, 20 to 35% of patients receiving CTRT do not experience clinical benefit, yet are exposed to the risks of CTRT therapy and potential unnecessary delay of surgery.
Knowing which patients are likely to experience extreme resistance to CTRT (ex-CTRT) would enable physicians to personalize treatment plans for these patients by considering carefully the risk of regimens to which response is unlikely.
Understand Individual Potential for CTRT Benefit
Test analyzes tumor biology to identify extreme resistance to CTRT
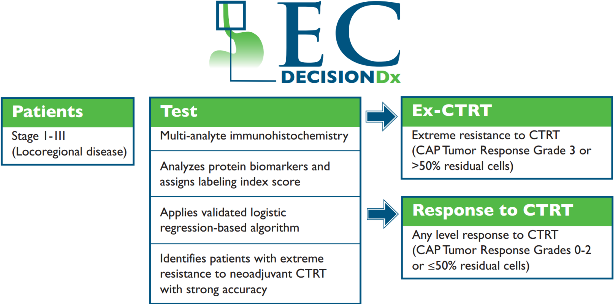
DecisionDx-EC accurately identifies patients who have likelihood of extreme resistance to neoadjuvant chemoradiation therapy by analyzing individual tumor biology. Using validated multi-analyte immunohistochemisty techniques, the test analyzes three protein biomarkers, NFkB, GLI1, and sonic hedgehog (SHH), in pre-treatment esophageal adenocarcinoma tissue. These pathways are known to mediate resistance to radiation and chemotherapy. Each biomarker is given a labeling index score based upon localization of the protein and compared to an established training set of samples with known outcomes using a validated logistic regression-based algorithm.
The test stratifies patients into two distinct classifications:
1) Ex-CTRT: Extreme resistance to neoadjuvant chemoradiation therapy (College of American Pathologists Tumor Regression Grade 3 or >50% residual tumor); or
2) Response to CTRT: Any level of response to neoadjuvant chemoradiation therapy (College of American Pathologists Tumor Regression Grades 0-2 or ≤50% residual tumor).
Potential Clinical Benefits
Predicting extreme resistance helps with treatment planning
Knowing which patients are likely to experience extreme resistance to pre-operative chemoradiation therapy allows patients unlikely to experience benefit from CTRT to avoid the potential morbidities associated with these treatment regimens and perhaps accelerating consideration of surgical intervention.
Reimbursement
Castle Biosciences works with Medicare, commercial insurers and the physician’s institution to secure payment coverage for DecisionDx-EC on the patient’s behalf. Insurance submission procedures may require Castle to receive relevant medical record and documentation support from the treating physician.
The Company also has put in place a generous Patient Assistance Program with the belief that care should not be contingent on financial considerations.
Select References
National Comprehensive Cancer Network (NCCN).
NCCN Clinical Practice Guidelines in Oncology.
Esophageal and Esophagogastric Junction Cancers Version 2.2013. 2013;
National Comprehensive Cancer Network.
van Hagen, et al. Preoperative chemoradiotherapy for esophageal or junctional cancer. N Engl J Med, 2012; 366: 2074-2084.
Sims-Mourtada, et al. Sonic Hedgehog promotes multiple drug resistance by regulation of drug transport. Oncogene, 2007; 26: 5674-5679.
Izzo, et. al. Diseases of the Esophagus, 2009.
Izzo, et al. Pretherapy nuclear factor-kappaB status, chemoradiation resistance, and metastatic progression in esophageal carcinoma. Molecular Cancer Therapeutics, 2006; 5: 2844-2850
Sims-Mourtada, et al. Hedgehog: an attribute to tumor regrowth after chemoradiotherapy and a target to improve radiation response. Clinical Cancer Research 2006; 12: 6565-6572.
Ajani, et al. Validated biomarker signatures that predict pathologic response to preoperative chemoradiation therapy (CTRT) with high specificity and desirable sensitivity levels in patients with esophageal cancer (EC). J Clin Oncol 29, 2011 (suppl; abstr 4027).
Rohatgi, et al. Failure patterns correlate with the proportion of residual carcinoma after preoperative chemoradiotherapy for carcinoma of the esophagus. Cancer 2005; 104 (7): 1349-1355.
Rohatgi, et al. Characterization of pathologic complete response after preoperative chemoradiotherapy in carcinoma of the esophagus and outcome after pathologic complete response. Cancer 2005; 104 (11): 2365-2372.
Dittrick, et al. Pathologic nonresponders after neoadjuvant chemoradiation for esophageal cancer demonstrate no survival benefit compared with patients treated with primary esophagectomy. Annals of Surgical Oncology 2012; 19: 1678-1684.
Lassen, et al. Clinical development of a proprietary immunohistochemistry (IHC) assay to detect pathologic complete response (pathCR) in esophageal adenocarcinoma (EC). Gastrointest Endosc 2013; 77 (suppl; abstr Su1526).